Table of Contents
How a Water Softener Uses Salt to Remove Hardness from Water
A water softener that uses salt for water softening removes lime and hardness from water through a process called ion exchange. In this process, calcium and magnesium ions—the minerals responsible for limescale—are removed from the water and replaced with harmless sodium ions.
Here’s how it works: tap water flows through the water softener and passes over resin granules inside the unit. These resin granules act like a magnet, attracting and capturing calcium and magnesium ions. Over time, the resin becomes saturated and needs to be regenerated.
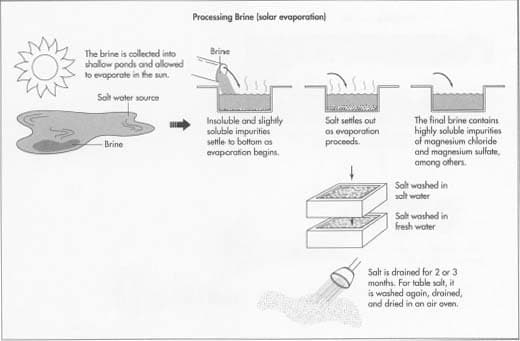
This is where regeneration salt comes into play. Adding salt to the water softener creates a concentrated brine solution, which rinses the resin granules. During this regeneration process, the captured calcium and magnesium ions are flushed away, restoring the resin’s ability to soften water efficiently.
With regular use of salt in your water softener, your system continues to provide soft, scale-free water for your home or business, protecting appliances, pipes, and improving overall water quality.