What Is the Chemical Formula for Salt?
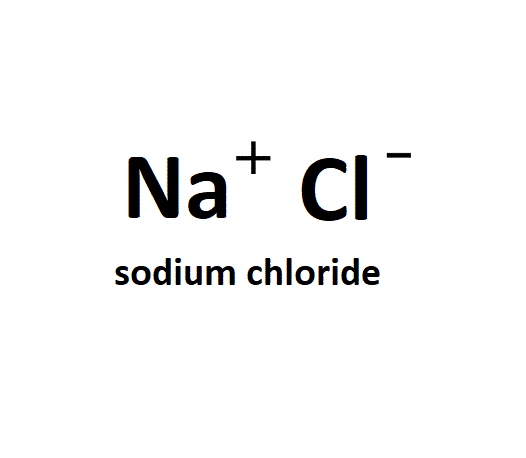
Salt is a common term used in everyday life, but chemically, it refers to a broad category of compounds. Understanding what salt is and the chemical formula for salt helps clarify its composition and properties. This article explains the basics of salt chemistry, including common compounds known as salts and their relevance in industry.
What Is Chemical Salt?
In chemistry, a salt is an ionic compound composed of positively charged ions (cations) and negatively charged ions (anions), which combine to form a neutral substance. Salts are typically formed by the reaction of an acid and a base.
Chemical Compounds for Salt
- The chemical formula for salt is NaCl.
- It consists of sodium (Na⁺) and chloride (Cl⁻) ions.
- Sodium chloride is widely used in food, water softening, industrial processes…
Other Examples of Salts
- Calcium chloride (CaCl₂): Often used as ice melt on roads.
- Potassium chloride (KCl): Used in fertilizers and water softeners.
- Magnesium sulfate (MgSO₄): Known as Epsom salt, used in agriculture and medicine.
Applications of Different Salts
- Water softening salt typically refers to sodium chloride or potassium chloride used to remove hardness from water.
- Ice melt salts often contain calcium chloride or sodium chloride due to their melting properties at low temperatures.
- Salts also play a crucial role in chemical manufacturing, food preservation, and environmental control.
Contact Us for Salt Supply
We offer worldwide logistics and tailored solutions to meet your needs efficiently and reliably.